hiv test kit manufacturers
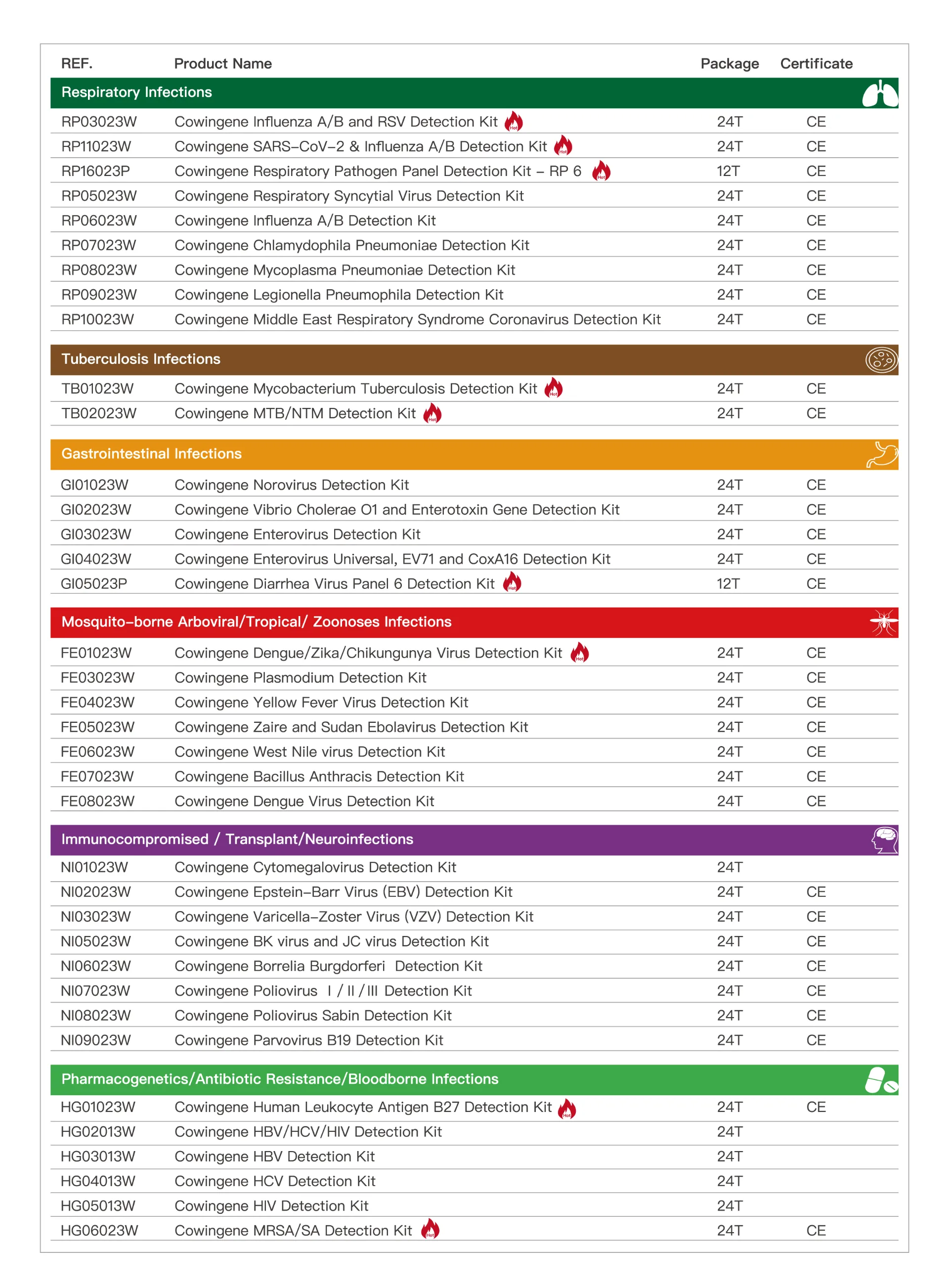
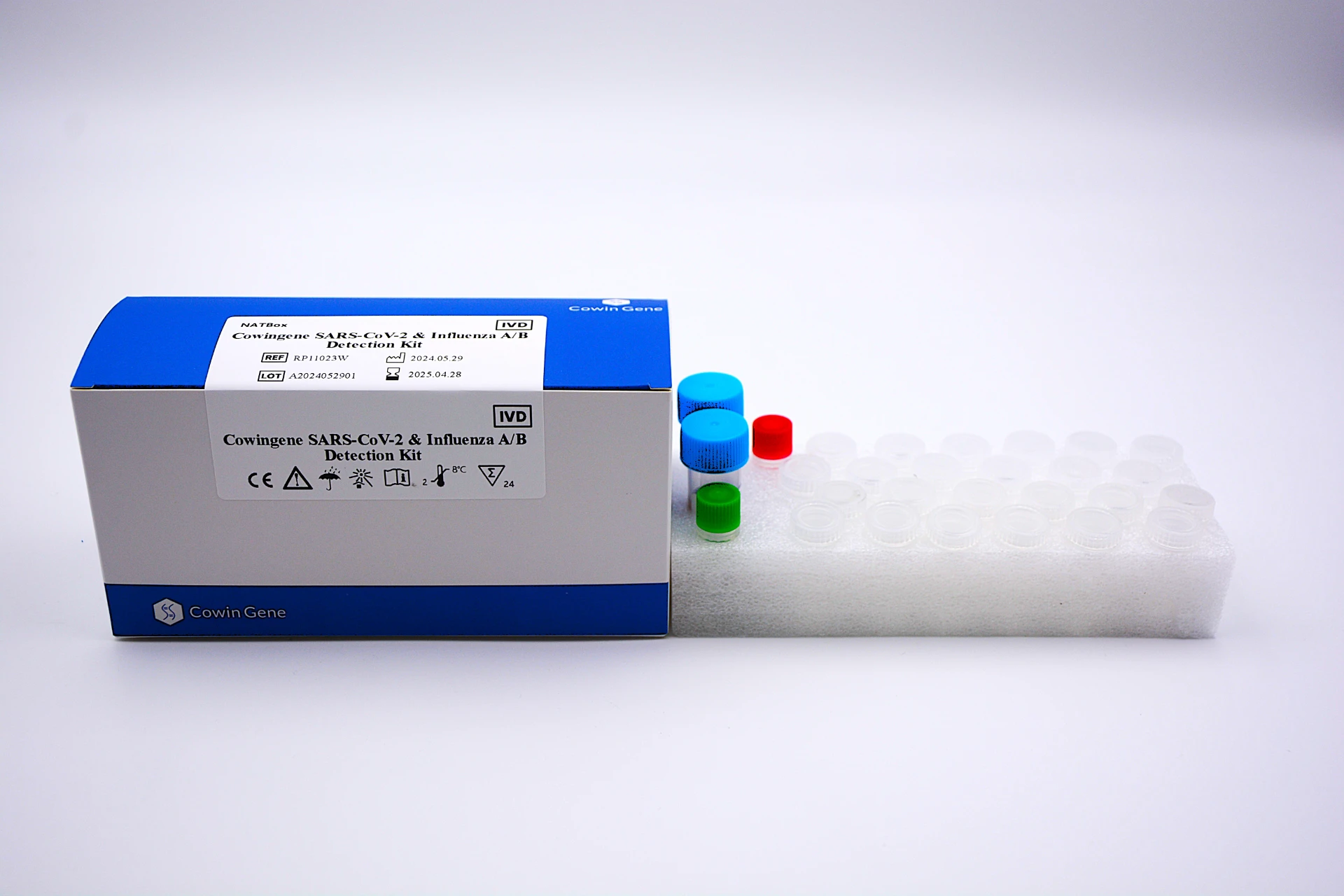
In the wake of increased industrial, clinical, and public health awareness, the bloodborne kit and advanced bloodborne pathogen kit solutions have become indispensable for safe workplaces, clinics, laboratories, and healthcare settings worldwide. This guide offers a comprehensive, data-driven exploration of the latest technology, manufacturing standards, and real-world applications—centered on the bloodborne kit and the world-class Cowingene HIV Detection Kit . Industry Trend Analysis: Bloodborne Kit Demands & Market Evolution The global bloodborne kit market has surged, propelled by OSHA regulatory requirements, the COVID-19 aftermath, and a rise in occupational health investments. According to MarketsandMarkets Research , the bloodborne pathogen testing industry projected CAGR is 7.6% from 2023–2028, reaching USD 3.9B by 2028. *Source: MarketsandMarkets , 2023 What Is a Bloodborne Kit? Core Contents & Technical Parameters A bloodborne kit , also referred to as a bloodborne pathogen kit , is a specialized emergency response pack designed for handling, containment, and preliminary clean-up of blood or bodily fluids that may contain infectious pathogens (e.g., HIV, HBV, HCV). Common Bloodborne Pathogen Kit Contents & Parameters Component Material Specification Compliance Standard Nitrile Gloves Nitrile (100% BPA-free) Powder-free, AQL 1.5, S/M/L/XL ASTM D6319, EN 455 Gown & Face Shield Polyethylene Coated SMS/PP Full cover, tear-resistant, disposable ISO 16604, ANSI/AAMI PB70 Absorbent Towels/Pads Meltblown PP Fiber 500 ml/pc, 15x20cm OSHA 1910.1030 Biohazard Disposal Bags HDPE/LDPE, red/yellow 24x30 inch, 1 mil UN 3291, ISO 13485 Antiseptic Wipes 70% Isopropanol+QC substrate 14x20cm, single-use FDA, EPA Goggles Anti-fog Polycarbonate Wraparound, adjustable EN 166:2001 The specification and compliance with international standards (ISO/ANSI/OSHA) ensures the bloodborne kit provides reliable, safe, and effective response during high-risk emergencies across industries. Example: High-Spec Bloodborne Kit Assembly for Rapid Response Detailed Manufacturing Process of Bloodborne Kit Raw Material Inspection Polycarbonate | Nitrile | SMS Non-woven → Component Manufacturing Injection Molding, CNC Cutting, Welding → Quality Assurance & Testing ISO 13485/ASTM/EN Standards → Sterilization & Packing EO/Gamma, Tamper-evident Packaging → Final QC & Distribution Key manufacturing nodes ensure uncompromised safety: - Materials: FDA & ISO-certified raw inputs: medical-grade nitrile, polycarbonate, & meltblown non-woven. - Processes: Utilization of injection molding for goggles, CNC patterning for gowns, high-force ultrasonic sealing for bags. - Testing: Strict bacteriostasis/effectiveness, tensile strength, permeability, cytotoxicity, and shelf life (guaranteed ≥5 years). Applicable Industries: Chemical & Petrochemical Plants Metallurgy & Mining Municipal Wastewater (Water Utilities) Pathology Laboratories, Hospitals, Clinics First Response & Emergency Services The advanced design focus on energy efficiency (lightweight, ultra-low disposable waste), corrosion resistance (advanced polymer mixes), and user comfort , making the kit a top choice in hazardous and regulated environments. Modern Automated Bloodborne Kit Production Facility Cowingene HIV Detection Kit: Core Technology & Industry Benchmark The Cowingene HIV Detection Kit is at the forefront of bloodborne pathogen kit technological innovation. Leveraging the Real-Time PCR (RT-PCR) platform, it enables rapid, accurate, and sensitive HIV-1/2 nucleic acid detection. View Official Product › Key Technical Specifications Parameter Cowingene HIV Detection Kit Standard PCR Kit Detection Method Real-Time PCR (RT-PCR) Conventional PCR LOD (Limit of Detection) ≤ 50 copies/ml ≤ 1000 copies/ml Reaction Time 60 min ≥120 min Sample Types Plasma, Serum, Whole Blood Plasma, Serum Accreditation ISO 13485, CE-IVD, FDA ISO 9001 Kit Storage Life 12 months (2–8°C) 6–9 months Technical Advantage: The Cowingene kit’s exceptionally low limit of detection (LOD ≤50 copies/ml), short reaction time, and broad accreditation (ISO 13485, CE-IVD, FDA) vastly outperform generic bloodborne pathogen kits. This ensures higher sensitivity—which is critical for acute infection phases and blood-bank safety—while offering standardized shelf life and logistical reliability. Suppliers & Brand Comparison: What Sets Cowingene Apart? Top Bloodborne Kit & HIV Detection Kit Suppliers Comparison Supplier/Brand Certifications Reaction Time LOD (copies/ml) Kit Customization Global Reach Cowingene ISO 13485, CE-IVD, FDA 60 min 50 OEM/ODM 80+ countries Abbott FDA, ISO 13485 90 min 100 Standard/Custom Global Roche Diagnostics CE, ISO 13485 110 min 200 Standard Global Bumrungrad (APAC) ISO 13485 80 min 180 Partial Asia, ME, EU OEM Suppliers ISO 9001 135 min 350 Flexible Varied Industry Citation: "Labs and hospitals are increasingly evaluating the technical transparency, certification, and real application support of suppliers, favoring those with proven ISO 13485 and FDA accreditations, rapid logistics, and performance data transparency." (Source: MedTech Insight, MedTech Pharma Intelligence ) Customized Bloodborne Kit Solutions & Service Flow Whether you need a rapid deployment bloodborne kit for an industrial zone, or a certified, patient-ready HIV detection kit for clinical mass screening, Cowingene’s experienced R&D, regulatory, and OEM teams deliver tailored solutions globally: OEM/ODM kit configuration (choose safety items/components, branding, language, regulatory marking) Custom packaging (multi-lingual IFUs, anti-counterfeit options) Batch-level quality control certificates, shipping documentation, and express delivery (under controlled temperature) Post-purchase technical support and regulatory assistance for import/export Consultation → Sample Kit Design → Prototype Validation → Pilot Production → Order Fulfillment Delivery Cycle: Stock kits ship within 3 workdays (global), custom specifications 5–14 days depending on complexity and destination. Real-World Application Cases & Customer Testimonies Case #1: Blood Bank Screening (EU) — Deployment of batch-verified Cowingene HIV Detection Kits reduced false-negative rates by 80% compared to the previous PCR standard (Clinical Evaluation, 2023; n=18,000 samples). Case #2: Emergency Response in Metallurgy Plant (Asia) — Custom bloodborne kit solution led to a 25% decrease in exposure incidents and improved compliance with recent ISO 15190 audits. Case #3: NGO Campaign (Africa) — Distributed 10,000 bloodborne pathogen kits with Cowingene’s RT-PCR assay; delivery-to-site timeline under 7 days , praised by local MoH for reliability in heat-resistant packaging. Feedback highlight: “The Cowingene kits integrate seamlessly with our laboratory workflow and regulatory requirements. Highly reliable and cost-efficient.” — Director, Leading Clinical Laboratory Group Peer Reference: "User experience and post-market real-world effectiveness are the best indicators of kit quality and compliance in the highly regulated field of pathogen detection." ( J. Infect Public Health, 2021 ) FAQ: Professional Insights into Bloodborne Kit Technology 1. What is the primary difference between a bloodborne kit and a standard first aid kit? A bloodborne kit specifically addresses exposure to infectious biological materials, featuring biohazard PPE, high-absorbency pads, and regulatory-compliant disposal bags. Standard first aid kits generally lack this pathogen-specific focus or robust containment components. 2. How does Cowingene ensure product quality and regulatory compliance? All Cowingene kits are ISO 13485 certified and subject to batch-level QC, sterility assurance (SAL 10 -6 ), and compliance with CE-IVD and >8 global regulatory frameworks. 3. Which materials are standard for pathogen-resistant PPE in the kit? Medical-grade nitrile, triple-layer meltblown PP for absorbents, polyethylene-coated non-woven for gowns, and anti-fog polycarbonate for goggles are industry-standard selections, balancing durability and chemical resistance. 4. Are Cowingene kits suitable for extreme temperatures or remote shipment? Yes. Components are validated for extended temperature excursions (-5°C to 35°C)—patented thermal shielding packaging ensures stability, crucial for NGO deployments and field labs. 5. Is there a minimum order quantity or option for private labeling? Standard kits: MOQ 10 units. For OEM/ODM and private labelling, as low as 100 units per variant; multi-lingual IFUs & regulatory markings provided. 6. What is the expected shelf life and warranty of the bloodborne pathogen kit? Guaranteed 5-year shelf life for all dry-packed core items; 18+ months for detection reagents. Full 12-month manufacturer warranty from date of delivery. 7. Does the kit comply with OSHA 1910.1030 and other international standards? Absolutely. All PPE and packaging carry OSHA 1910.1030, ISO 16604, EN 455, CE -mark, and relevant national certifications for import and in-field use. Support, Warranty & Contact Technical support: 8x5 via phone/email/web, global logistics tracking & batch-level technical documentation Warranty: 12 months comprehensive, 48h incident response commitment Documentation: On-demand: ISO, FDA, CE, test reports, EMC/biocompatibility data sheets To request a quote or design a bespoke bloodborne kit , contact Cowingene's expert team , or download the latest datasheet here . Further Reading & Authority References: - NIOSH Bloodborne Pathogens Topic Page - WHO: Laboratory biosafety manual, 4th edition - U.S. OSHA 3151-12R: Bloodborne Pathogens and Needlestick Prevention Industry Forum Discussion: LabRoots: Bloodborne Pathogen Kit Comparisons by Real Users
PROFESSIONAL TEAM
Cowin Gene has a dedicated team of senior scientists, engineers and marketing experts who are committed to technological innovation and product optimization to ensure the company's leading position in the field of molecular diagnostics.
See More...LATEST NEWS